diabetes
exhaled breath isoprene rose significantly at hypoglycemia compared with nonhypoglycemia." (A hypoglycemic attack occurs when blood sugar decreases to dangerous levels).
 organic compound produced by many plants and, to a lesser extent, humans (produced at higher rate in males). It has a faint hydrocarbon odor (rotten eggs) and gives its characteristic smell to hot rubber. Also known as isoterpene, 2-methyl-1,3-butadiene, Isopentadiene and 2-Methylbutadiene. diabetes exhaled breath isoprene rose significantly at hypoglycemia compared with nonhypoglycemia." (A hypoglycemic attack occurs when blood sugar decreases to dangerous levels).
Comments
 phytochemical, "spicy" volatile oil found in tangy herbs such as horseradish, mustard, and onions. Also called " mustard oils", many natural isothiocyanates from plants are produced by enzymatic conversion (myrosinase-mediated hydrolysis) of metabolites called glucosinolates - sulfur-containing compounds found in cruciferous vegetables. Examples: Allylisothiocyanate, Benzyl Isothiocyanate, Phenyl Isothiocyanate, Sulforophane. Isothiocyanates, such as sulforaphane, may help prevent cancer by promoting the elimination of potential carcinogens from the body and by enhancing the transcription of tumor suppressor proteins. The isothiocyanates with the stongest anticancer effects are phenylethylisothiocyanate, benzylisothiocyanate and 3-phenylpropylisothiocyanate. Studies have shown that isothiocyanates help to prevent lung cancer and esophageal cancer. Isothiocyanates can also lower the risk of other cancers, including gastrointestinal cancer. Several patented applications propose to use Isothiocyanates as pesticides, and as supplements in the diet to help prevent cancer. 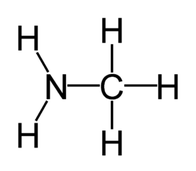 Simplest biogenic amine, derivative of ammonia with one H atom replaced by a methyl group. Has with a strong odor resembling rotten fish Methylamine and dimethylamine may be formed as metabolites of basic drugs containing N-methyl group. Can be detected in human breath and increased in certain conditions: e.g. because of amine catabolism increase in diabetes. It has been reported that transient irritation of the eyes, nose, and throat has resulted from brief exposures to MEthylamine concentrations of 20 to 100 ppm; the odor was intolerable at 100 to 500 ppm. Inhalation of methylamine vapors at concentrations greater than 100 ppm has caused irritation of the nose and throat, followed by violent sneezing, burning sensation of the throat, coughing, constriction of the larynx and difficulty in breathing, pulmonary congestion, and edema of the lungs. 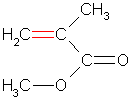 Organic compound with the formula CH₂=CCOOCH₃, CAS 80–62–6 Used in n chain-addition polymerizations, in the manufacture of methacrylate resins and plastics (e.g., Plexiglas; advertising signs and displays, lighting, plumbing and bathroom fixtures; lacquer, and enamel resins; concrete additives, and prosthetic medical devices - such as hip and knee joint prostheses). It can be detected in small concentrations in human breath - Streptococcus pneumonia infections, for example, increase its concentrations up to 1420 ppb. Methyl methacrylate is a flammable and reactive chemical, fire and explosion hazard. Breathing it in can irritate the lungs causing coughing and or shortness of breath. Higher exposures can cause a build up of fluid in the lungs and pulmonary edema. It can affect liver and kidneys, damage nervous system, causing numbness, “puns and needles”, and/or weakness in the hands and feet, and may cause a skin allergy. High exposure can cause dizziness, irritability, difficulty with concentration and reduced memory. Workplace exposure limits are 100 ppm over 8 hrs shifts or 50 ppm if longer term exposures. 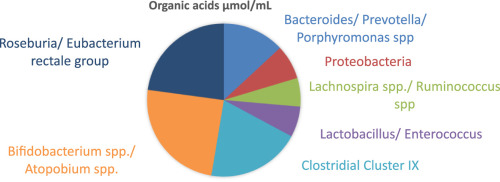 Organic compounds with acidic properties. Organic acids are considered to be any organic carboxylic acid of the general structure R-COOH, including fatty acids and amino acids. Short-chain organic acids (C1–C7) are widely distributed in nature as normal constituents of plants or animal tissues. They are also formed through microbial fermentation of carbohydrates mainly in the large intestine. They are sometimes found in their sodium, potassium, or calcium salts, or even stronger double salts. For optimal digestive health, organic acids need to be active throughout the length of the digestive tract. 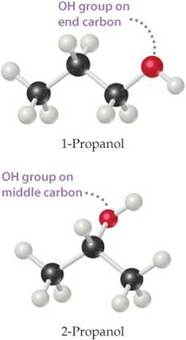 alcohols with a sharp musty sweet chemical odor like rubbing alcohol There are two isomers of propanol:
1-Propanol is metabolized by alcohol dehydrogenase to propionic acid (vinegar-like smell) and presents with metabolic acidosis and elevated anion gap, whereas 2-propanol is metabolized by alcohol dehydrogenase to acetone (fruity, nail-polish-like smell) and presents with rapidly developing (within 3-4 h after exposure) ketosis and ketonuria but without metabolic acidosis. 2-propanol smells more like rubbing alcohol (Rubbing alcohol, hand sanitizer, and disinfecting pads typically contain a 60–70% solution of isopropyl alcohol in water) resembling more of acetone, and could be hazardous to health. Propanol in breath might correlate with acetone. It is present in the breath samples of cystic fibrosis (CF) patients infected by Pseudomonas aeruginosa (PA). There could be multiple sources of propanol. For example, propane-utilizing bacterium Gordonia sp. strain TY-5, oxidizes propane gas to 2-propanol that is further oxidized to acetone. 1-propanol is a by-product formed from certain amino acids when potatoes or grains are fermented to produce ethanol. 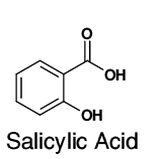 bitter acid present in plants, used in making aspirin and as a preservative. Also known as orthohydroxybenzoic acid and Saicylate, this organic acid is named after white willow tree (salix), from the bark of which it is used to be obtained. Wintergreen leaves are also high in Salycilate. Possesses bacteriostatic, fungicidal, and keratolytic action, used as a food preservative, an antiseptic in toothpaste, a peeling agent in ointments, creams, gels, and shampoos applied to reduce the scaling of the skin or scalp in psoriasis, in skin products for the treatment of acne since it causes skin cells to slough off more readily, preventing them from clogging up the pores. Highest content in Salycilate drugs like Aspirin, in food spices such as cumin, paprika, turmeric, dry dill, oregano, rosemary, thyme, mustard, curry powder, especially Vindaloo curry. Relatively high in some mushrooms, olive oil, apricots. Salicylic acid is a metabolite of these foods and drugs. 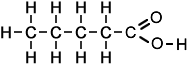 Straight-chain alkyl carboxylic fatty acid with a penetrating unpleasant fatty-earthy odor, acidic and sharp, old cheese-like, sour-milky, sweaty, rancid, resembling tobacco with fruity nuances. One of SFCAs (n=6). Molecular formula: C5H10O2 Found naturally in the perennial flowering plant valerian (Valeriana officinalis). Volatile esters of valeric acid tend to have pleasant fruity odors and are used in perfumes and cosmetics. Valeric acid can cause irritation if it comes into contact with the skin, eyes, or mucous membranes. It can be detected in human blood, feces, saliva and urine as Lactobacilli, bifidobacteria, clostridia, and enterococci secrete valerate as well as other acids including acetate, propionate, butyrate, and lactic acid. 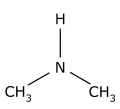 Also known as N-Methylmethanamine and DMA, this biogenic (aliphatic) amine can be detected in the urine of humans and other mammals; it is produced in the metabolism of choline and choline-containing phosphatides and has strongly ammoniacal odor that could be also fish-like. Urinary excretion in humans is increased after eating certain fish, meat, dairy, and grains containing dimethylamine. The German cockroach (smaller species) utilizes dimethylamine as a pheromone for communication. Dimethylamine is used in leather tanning and as an accelerator in the manufacturing of rubber, detergents, and pesticides. Odor threshold is 1.6 ppm, irritation may occur at 5 ppm or higher. Exposure Routes are via inhalation, skin and/or eye contact. Symptoms range from irritation of nose and throat; sneezing, cough, dyspnea (breathing difficulty); pulmonary edema; conjunctivitis; dermatitis; liquid: frostbite. Because of its good solubility in lipids, dimethylamine is readily and rapidly absorbed in the gastrointestinal and respiratory tracts and acts as a central nervous system depressant. Concentrated dimethylamine vapors are highly irritating to mucous membranes. There is no evidence of carcinogenic potential. 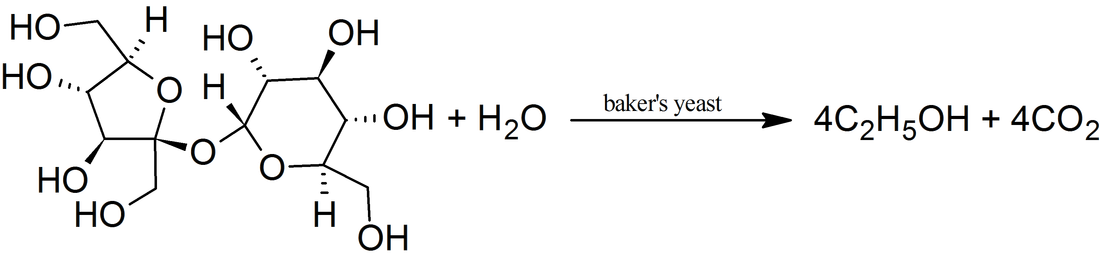 ethyl alcohol, also known as grain alcohol (absolute alcohol if undiluted), is the principal type of alcohol found in alcoholic beverages. This compound with molecular formula C2H5OH has a very distinctive perfume-like smell. It is produced by the fermentation of sugars by yeasts, most notably, Saccharomyces cerevisiae. Ethanol can be measured in breath (30 minutes after consumption), sweat (over 40 minutes after consumption) and blood. The presence of ethanol in blood at concentrations >30 mg/dL (>0.03% or g/dL) is generally accepted as a strong indicator of the use of an alcohol-containing beverage. Ethanol can be also detected in high amounts in the blood of those with auto-brewery syndrome (gut fermentation syndrome) or smaller but still elevated amounts with yeast or bacterial dysbiosis (overgrowth) through endogenous fermentation within the digestive system. |
Categories
All
|