
Acerola berries could possibly boost immune system and metabolism, be a remedy for diarrhea, anemia, diabetes, dysentery, fever, heart function. Cross-allergies with latex.
 juicy and soft cherry-like fruit with a pleasant, tart flavor. Grows on a tropical shrub/small tree in the family Malpighiaceae. Common names include Acerola , Barbados cherry, West Indian cherry and wild crepe myrtle.Acerolla is known for being extremely rich in vitamin C, but it also contains vitamins A, B1, B2, and B3, and antioxidants carotenoids and bioflavonoids. Acerola berries could possibly boost immune system and metabolism, be a remedy for diarrhea, anemia, diabetes, dysentery, fever, heart function. Cross-allergies with latex.
Comments
 Calorie-free sugar substitute (artificial sweeetener), potassium salt of 6-methyl-1,2,3-oxathiazine-4(3H)-one 2,2-dioxide. Also known as E950, acesulfame K, Ace K, Sunett and Sweet One, Acesulfame potassium is as sweet as aspartame, 180-200 times sweeter than sucrose, half as sweet as saccharin (that also has a slightly bitter aftertaste), and only a quarter as sweet as sucralose. Acceptable daily intake is 15 mg/kg body weight. Acetaminophen (United State Adopted Name: USAN) or Paracetamol (International Nonproprietory name: INN) is a popular drug used for the relief of fever, headaches, and other minor aches and pains. It's analgesic (pain reliver) and antipyretic (reducing fever). Acetaminophen is a major ingredient in numerous cold and flu medications and many prescription analgesics. It is safe in standard doses, but because of its wide availability, deliberate or accidental overdoses are not uncommon. Acetaminophen, unlike other common analgesics such as aspirin and ibuprofen, is not a non-steroidal anti-inflammatory drug or NSAID. In normal doses acetaminophen does not irritate the lining of the stomach nor affect blood coagulation, the kidneys, or the fetal ductus arteriosus (as NSAIDs can). Acetaminophen is used on its own or in combination with pseudoephedrine, dextromethorphan, chlorpheniramine, diphenhydramine, doxylamine, codeine, hydrocodone, or oxycodone. General Dosing Guidelines for adults: 325 to 650 mg every 4 to 6 hours or 1000 mg every 6 to 8 hours 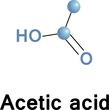 organic acid which gives vinegar its sour taste and pungent smell. Produced by acetic acid bacteria including Clostridium acetobutylicum and Acetobacter, found universally in food, water and soil, produced in food spoilage and in the human body - for example, in vaginal lubrication. This second simplest carboxylic acid (after formic acid) is an important chemical reagent and industrial chemical, mainly used in the production of cellulose acetate for photographic film and polyvinyl acetate for wood glue, as well as synthetic fibers and fabrics. the acid that gives vinegar its characteristic taste. One of short chain fatty acids (SCFAs, also called volatile fatty acids) containing fewer than 6 carbons: Formic acid (C1), acetic acid (C2), propionic acid (C3), butyric acid (C4), and valeric acid (C5). The gut microbial metabolites SCFAs profoundly regulate T cell differentiation in the body, boosting immune system. if raised, suggests bacterial fermentation due to excess carbohydrate reaching the colon; levels are not significantly altered in patients with malignancy or diabetes mellitus, but severe liver disease and severe acidosis were both associated with increased acetate concentrations. Elevated blood acetate is also an indicator of fast ethanol elimination in chronic alcoholics.  Acetobacter is bacteria converting alcohol to acetic acid and oxidizing lactate and acetate into carbon dioxide and water. It is used in the commercial production of vinegar, as fermentation starter cultures, and is found in gut microbiomes of some living organisms. Acetobacter aceti bacteria are a widespread group of gram-negative, obligate aerobic rods which oxidize ethanol (alcohol) into ethanoic (acetic) acid. They occur mainly in sugary, acidic and/or alcoholic surroundings and play a positive, neutral, or detrimental roles in food and beverages. It can be found in mangoes, bananas, sugar cane, sweet potato, coffee, cameroon grass, ragi, tea, pineapple, and mealy bugs. 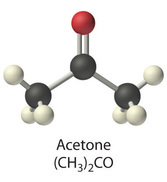 a colorless volatile liquid ketone with a fruity smell resembling nail polish remover or paint thinner. It is made by oxidizing isopropanol, and is used as an organic solvent, synthetic reagent, and as an antiseptic. Acetone occurs naturally in plants, trees, forest fires, vehicle exhaust and as a breakdown product of animal fat metabolism. It may be normally present in very small quantities in urine and blood; larger amounts may be found in the urine and blood of diabetics. Larger concentrations of acetone in breath are also associated with fasting and sinusitis. 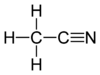 a clear, colorless liquid with a sweet burnt taste and sweetish ethereal odor, that might resemble sweaty feet. Formula CH3CN, CAS number 75-05-8 Acetonitrile vapors have been detected in feces, breath, blood and saliva of healthy individuals. At higher concentrations, it is a hazardous chemical substance and, if inhaled, can produce serious acute (short term) toxicity, including loss of consciousness or death. Toxicity depends on the amount, route (inhalation, contact, and ingestion), time and frequency of exposure(s); acute symptoms are usually abdominal pain, convulsions, labored breathing, weakness, unconsciousness and redness in the skin and eyes. With prolonged exposure, the liver, lungs, kidneys, and central nervous system may be affected. Environmental sources of acetonitrile include grass or other biomass burning and automobile exhaust. Acetonitrile poisoning can also occur by drinking contaminated water or by ingesting contaminated foods. 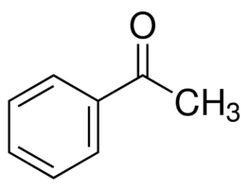 simplest aromatic ketone, in small amounts can be found in apple, apricots, cheese, chicory, banana, beef, and cauliflower. It is a precursor to useful resins and fragrances, is used in chewing gum and is an ingredient in fragrances that resemble smell of almond, cherry, honeysuckle, jasmine, and strawberry. In humans, acetophenone is metabolized to benzoic acid, carbonic acid, and acetone, but it can be also excreted in its original form in feces and saliva. Deuterated acetophenone smells fruitier than ordinary acetophenone. It also smells slightly of bitter almonds, just like many compounds containing the cyanide or nitrile group (C≡N) - as both C–D and C≡N bonds vibrate at the same frequency (about 2200 cm-1). See also 2-aminoacetophenone  smells associated with sour and acidic substances such as vinegar, fermenting milk or vegetables, wine or lemon. Can be pungent and sharp as Formic acid and Capric acid in goats’ milk, vinegary as acetic and propionic acid, or sourish and cheesy as isovaleric acid responsible for "locker room" odor, "dirty socks" smell and stinky feet. Some acidic breath or body odors are associated with yeast infections, cystic fibrosis and cancers, but it could also be a side effect of diet, medications and microbial imbalance (skin and gut flora) |
Categories
All
|