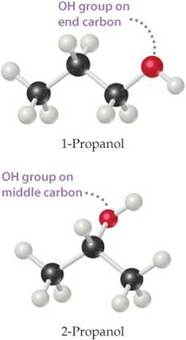
There are two isomers of propanol:
- 1-Propanol, n-propanol, propan-1-ol, ethylcarbinol
- 2-Propanol, Isopropyl alcohol, isopropanol, propan-2-ol
1-Propanol is metabolized by alcohol dehydrogenase to propionic acid (vinegar-like smell) and presents with metabolic acidosis and elevated anion gap, whereas 2-propanol is metabolized by alcohol dehydrogenase to acetone (fruity, nail-polish-like smell) and presents with rapidly developing (within 3-4 h after exposure) ketosis and ketonuria but without metabolic acidosis. 2-propanol smells more like rubbing alcohol (Rubbing alcohol, hand sanitizer, and disinfecting pads typically contain a 60–70% solution of isopropyl alcohol in water) resembling more of acetone, and could be hazardous to health.
Propanol in breath might correlate with acetone. It is present in the breath samples of cystic fibrosis (CF) patients infected by Pseudomonas aeruginosa (PA).
There could be multiple sources of propanol. For example, propane-utilizing bacterium Gordonia sp. strain TY-5, oxidizes propane gas to 2-propanol that is further oxidized to acetone. 1-propanol is a by-product formed from certain amino acids when potatoes or grains are fermented to produce ethanol.
Each of two isomeric liquid alcohols is used as solvents.
Vapors of propanol are heavier than air and mildly irritate the eyes, nose, and throat. Density approximately 6.5 lb / gal. Used in making cosmetics, skin and hair preparations, pharmaceuticals, perfumes, lacquer formulations, dye solutions, antifreezes, rubbing alcohols, soaps, window cleaners, acetone and other chemicals and products.
2 - Isopropanol is an isomer of propyl alcohol with antibacterial properties. Although the exact mechanism of isopropanol's disinfecting action is not known, it might kill cells by denaturing cell proteins and DNA, interfering with cellular metabolism, and dissolving cell lipo-protein membranes. Isopropanol is used in soaps and lotions as an antiseptic.
Isopropyl alcohol is used as a water-drying aid for the prevention of otitis externa, better known as swimmer's ear. Isopropyl alcohol and its metabolite, acetone, act as central nervous system (CNS) depressants. Symptoms of isopropyl alcohol poisoning include flushing, headache, dizziness, CNS, depression, nausea, vomiting, anesthesia, hypothermia, hypotension, shock, respiratory depression, and coma. Poisoning can occur from ingestion, inhalation, or skin absorption; therefore, well-ventilated areas and protective gloves are recommended.
Around 15 g of isopropyl alcohol can have a toxic effect on a 70 kg human if left untreated. However, it is not nearly as toxic as methanol or ethylene glycol.
1-propanol and 2-propanol poisoning presents early with mixed acidosis and elevated anion gap and only later with ketonuria.
2-Methyl-2-propanol is a metabolite of isobutane, used in fuels and aerosol sprays. The presence of this metabolite in blood samples confirms the antemortem presence of isobutane.
2,3-Dibromo-1-propanol is a metabolite of the flame retardant tris(2,3-dibromopropyl) phosphate. 2-(2-Pyridinyloxy)-1-propanol is a metabolite of the pesticide Pyriproxyfen.
