Coronary stents:
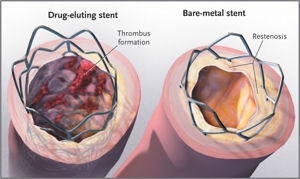
Bare-metal stents (BMS) were introduced in the mid-1990s with the aim of overcoming the deficiencies of balloon angioplasty (it could weakens the artery wall making it collapse once the balloon is deflated. Besides, In between 30 and 40 percent of all procedures within the first year, the artery begins to renarrow as a result of the body’s healing response to the trauma of angioplasty, the equivalent of scar tissue forming over an injury.). Subsequent refinement to earlier designs offer improved flexibility, making it easier to deliver to the narrowed artery, yet, in between 20 and 30 percent of cases the treated artery was found to re-narrow within six months of stent insertion, leading to the need for a repeat procedure. While the risk of developing in-stent restenosis is linked to a variety of clinical and procedural factors (particularly diabetes, long lesions, small vessels and procedural failure), all BMS, regardless of the thickness of the struts, provoke a considerable proliferative response.
Drug-eluting stents (DES) were developed to specifically address the problems of restenosis encountered with BMS. Some drugs, such as paclitaxel, can be coated directly on a metal stent, whereas the majority of the drugs need to be attached to a polymer, which acts as a drug reservoir
The first DES to be launched was the Cypher® stent in 2003, followed by the Taxus® stent in 2004. Cypher was eluting Sirolimus (rapamycin) - a sophisticated natural antibiotic, developed for its powerful immunosuppressive activity (SES). Taxus contained Paclitaxel (1 microg/mm) - Taxol®, a plant alkaloid. a drug used to treat ovarian, breast, lung, pancreatic and other cancers. These were first generation drug-eluting stents (1st generation DES): sirolimus-eluting stent (SES) and paclitaxel-eluting stents (PES).
2nd-generation DES, such as zotarolimus-eluting (Endeavor® - ZES), Endeavor®Resolute (ZES-R). and everolimus-eluting stents (Xience V® - EES), became available in the USA and/or Europe by 2007-2008, and the first-in-human clinical trials of these generation were completed in 2003. The clinical benefits of one DES over another are less clear-cut, although a number of studies have shown that second generation DES are superior to original DES in terms of preventing further cardiovascular complications or the need for a repeat stenting procedure, as with Xience® versus Cypher.
In 2009, it was reported that compared with the other DES, ZES appeared less effective than SES but better than PES at 3 years follow-up, as confirmed by the 3-year results of the ENDEAVOR IV trial, presented at the Transcatheter Cardiovascular Therapeutics (TCT) 2009 meeting by Martin Leon
A 2013 study named everolimus-ES and Resolute zotarolimus-ES emerging as the safest stents to date, while second-generation biodegradable polymer-based DES fell short of high expectations. Many complex and critically relevant, interplaying factors influence the individual clinical outcome, including lesion complexity, coronary anatomy, comorbidities, and operator technical skill.
A 2014 study concluded that second-generation EES and ZES-R offer similar levels of efficacy compared with first-generation SES, but are more effective than PES; however, only second-generation EES significantly reduced the incidence of heart attack (MI) and stent thrombosis (ST), and therefore should be perceived as the safest DES to date.
3rd generation DES started to appear commercially in 2010-2011.
While durable polymers and eluted drugs, such as everolimus and zotarolimus, are all components of the second generation DES, it is the advanced base metallic structure of these newer stents that justifies the category of third generation. Platinum Chromium is the foundation of the ION paclitaxel-eluting platinum chromium coronary stent system. Other examples of 3rd generation drug eluting stents include ‘Resolute Integrity’ that releases the drug zotarolimus and ‘Promus Element’ that releases the drug everolimus.
Biodegradable polymers have been developed to overcome the long-term adverse vascular reactions related to the durable polymer. Biolimus-eluting stent (BES) is using biodegradable polymers. And so are 3rd generation Orsiro Hybrid sirolimus-eluting stentd with biodegradable polymer (O-SES, Biotronik) vs the widely used 2nd generation zotarolimus-eluting stents with durable polymer.
However, and disappointingly, with respect to safety endpoints, second-generation biodegradable polymer-based DES fell short of high expectations, and differences when compared with second-generation DP DES become obvious, with everolimus-eluting stents and Resolute zotarolimus-eluting stents emerging as the safest stents to date.3
Newer third-generation stent technology, especially biodegradable polymers, polymer-free stents, and biodegradable stents on the basis of poly-L-lactide (PLLA) or magnesium, has been evaluated in preclinical and initial clinical trials. However, despite encouraging initial results, long-term data of large-scale randomized trials as well as registries comparing them to currently approved first- and second-generation DES are still lacking.
